
Michael Harrison, MD, widely regarded as the father of fetal surgery, originally planned on becoming a general practitioner like his father. “He was an old-fashioned country doctor who went on house calls and had wonderful relationships with his patients,” said Harrison.
But at Harvard Medical School, Harrison became entranced with surgery and made it his life’s path. During his general surgery internship at Massachusetts General Hospital, he assisted legendary pediatric surgeon W. Hardy Hendren, MD. One patient was born with congenital diaphragmatic hernia, a rare condition in which a hole in the diaphragm allows the abdominal organs to migrate into the chest cavity, inhibiting lung growth.
Although Hendren performed a flawless operation to repair the defect, the child died. “I was so naïve that I said, ‘It’s obvious that the kid died not because he had this anatomic problem [of a hole in the diaphragm], but because his lung didn’t have a chance to grow – and the only way to save that kid was to fix the problem before birth,’” recalled Harrison. “When I told Dr. Hendren, he was so shocked he almost fell down – because if you know enough, you just don’t think that way.”
Harrison thought about that problem for the rest of his surgical training, then spent two years at the National Institutes of Health investigating why the mother does not reject the fetus – a foreign organism with a different immune system. “I thought, ‘I’ll just solve that problem,’ because that would be the answer to all of transplantation, which was the big, sexy thing in surgery then,” he said. Using animal models, he also started to develop techniques for fetal surgery.
Pioneering Fetal Surgery
In 1978, Harrison joined the UCSF faculty to continue his pursuit of fetal surgery – one of the few institutions nationally with the resources and open-mindedness to consider such a bold idea. Several UCSF scientists were studying cardiac development in animal models, and he wanted to partner with them. Also, radiologist Roy Filly, MD, and his group were pioneering the use of fetal ultrasound to diagnose anatomical defects in utero.
 Harrison forged relationships with other specialists, attending a standing meeting every Tuesday at 1 p.m. with obstetricians, pediatricians and other providers coordinating the care of pregnant women and newborns. Occasionally, the group would discuss fetuses with abnormalities such as congenital diaphragmatic hernia.
Harrison forged relationships with other specialists, attending a standing meeting every Tuesday at 1 p.m. with obstetricians, pediatricians and other providers coordinating the care of pregnant women and newborns. Occasionally, the group would discuss fetuses with abnormalities such as congenital diaphragmatic hernia.
“I’d say, ‘We could think about fixing that before birth, because that’s a really bad problem and he’s not going to live otherwise,’ and everyone said, ‘What? Are you crazy?’” recalled Harrison. “But because everyone saw the whole course of the disease – the baby was delivered and died – the next time they said, ‘Maybe it makes sense to do something.’
Harrison worked with those colleagues, as well as bioethicists and the Institutional Review Board, to wrestle with the complex ethics of fetal surgery. They determined that initial cases must address severe conditions certain to prove lethal if untreated before birth. Because each fetal surgery has two patients – the mother and the fetus – they agreed that if an intervention ever injured a mother, they would discontinue fetal surgery.
Harrison’s team also performed hundreds of fetal surgeries on monkeys at UC Davis’s California National Primate Research Center, developing a technique to safely open and close the uterus in mid-gestation. When none of the monkeys could become pregnant again, they realized that the metal staples acted as an intrauterine device. Harrison worked with the manufacturer to develop absorbable staples, which are still used today.
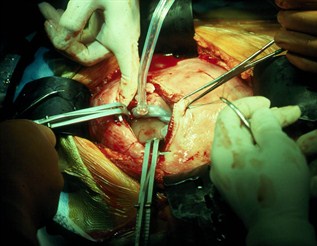
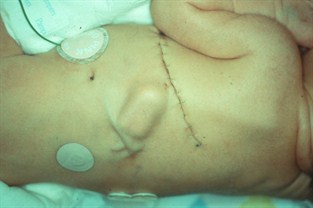 Harrison and his colleagues performed the first open fetal surgery in 1981 to unblock a urinary tract obstruction. The surgery accomplished its goal, but the child died after delivery because the intervention happened too late – after the kidneys had already failed and the lack of amniotic fluid prevented adequate lung development. Later that year, they performed a successful case, using a minimally invasive approach and inserting a Harrison vesicoamniotic shunt – a device that Harrison invented which uses a tube to connect the obstructed bladder with the amniotic space. That fetal patient required an additional corrective procedure after birth, but today is a healthy adult.
Harrison and his colleagues performed the first open fetal surgery in 1981 to unblock a urinary tract obstruction. The surgery accomplished its goal, but the child died after delivery because the intervention happened too late – after the kidneys had already failed and the lack of amniotic fluid prevented adequate lung development. Later that year, they performed a successful case, using a minimally invasive approach and inserting a Harrison vesicoamniotic shunt – a device that Harrison invented which uses a tube to connect the obstructed bladder with the amniotic space. That fetal patient required an additional corrective procedure after birth, but today is a healthy adult.
Harrison and Filly co-founded the Fetal Treatment Center at UCSF, the birthplace of fetal surgery, which has more experience in this field than any other institution in the world. The Fetal Treatment Center team continues to meet every Tuesday at 1 p.m., convening about 50 experts from diverse fields to develop specialized treatment plans for each patient.
Over the years, Harrison and his colleagues developed less invasive methods for performing fetal surgery to reduce the risk of preterm labor, the leading complication of fetal surgery. They also pioneered interventions for many congenital abnormalities, including spina bifida, twin pregnancy complications, congenital pulmonary airway malformation, and many other disorders.
Creating New Tools
 The team also developed new devices to improve and refine fetal interventions. “We had to make up tons of things for fetuses, because there was nothing,” said Harrison. He and his colleagues would meet at 5 a.m. before their first surgical cases of the day to brainstorm new ideas. Even after retiring from surgical practice in 2006, Harrison continued to develop new devices. With a grant from the US Food and Drug Administration (FDA), he established the UCSF Pediatric Device Consortium – affectionately nicknamed the “D’Vice Squad” – to bring together inventors with a shared passion for device development.
The team also developed new devices to improve and refine fetal interventions. “We had to make up tons of things for fetuses, because there was nothing,” said Harrison. He and his colleagues would meet at 5 a.m. before their first surgical cases of the day to brainstorm new ideas. Even after retiring from surgical practice in 2006, Harrison continued to develop new devices. With a grant from the US Food and Drug Administration (FDA), he established the UCSF Pediatric Device Consortium – affectionately nicknamed the “D’Vice Squad” – to bring together inventors with a shared passion for device development.
The Pediatric Device Consortium’s philosophy is similar to the interdisciplinary approach that led to the Fetal Treatment Center, with its regular meetings of specialists with complementary expertise. “We meet twice a week, and bring together 10 or 20 people of wildly different background – pediatric surgeons, a cardiologist, an engineer from San Francisco State, mechanical and electrical engineers,” said Harrison. “Anybody can come to the meeting, and there’s no holding back – no one worries, ‘That’s my idea!’” Meetings include guest lectures and brainstorming sessions.
Harrison and his colleagues are developing many new devices, including:
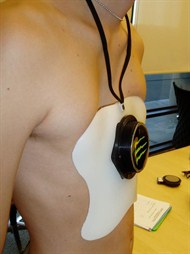 Magnetic mini-mover: Pectus excavatum, also known as sunken chest disorder, is one of the most common chest wall deformities. It can only be repaired through a major operation requiring up to seven days of hospitalization. “The kids are in agony for a week, and it’s a brutal operation – I hated it every time I did it,” said Harrison. “I wondered how we could do it better, and thought – we need to pull on the chest gradually, rather than trying to fix it in one giant operation.”
Magnetic mini-mover: Pectus excavatum, also known as sunken chest disorder, is one of the most common chest wall deformities. It can only be repaired through a major operation requiring up to seven days of hospitalization. “The kids are in agony for a week, and it’s a brutal operation – I hated it every time I did it,” said Harrison. “I wondered how we could do it better, and thought – we need to pull on the chest gradually, rather than trying to fix it in one giant operation.”
With the Magnetic Mini-Mover Procedure, surgeons implant a magnet in the sternum, usually in an outpatient procedure. The patient wears an orthotic brace with a second magnet that gradually reforms the chest wall to a normal position. The device is currently completing a Phase III multi-center clinical trial.
- Magnap: Obstructive sleep apnea occurs when the airway becomes narrowed or blocked during sleep. If left untreated, it can contribute to high blood pressure, heart failure or stroke. A continuous positive airway pressure (CPAP) machine can effectively treat the condition, but many patients dislike the uncomfortable masks and noisy pumps.
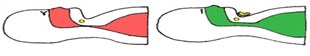 With Magnap, surgeons implant a small magnet encased in titanium on a patient’s hyoid bone, which is attached to soft tissue in the neck that blocks the airway in obstructive sleep apnea. Just before bedtime, patients place a plate with a second magnet above the throat area, which opens up the airway. The Magnap group is currently recruiting patients for a clinical trial.
With Magnap, surgeons implant a small magnet encased in titanium on a patient’s hyoid bone, which is attached to soft tissue in the neck that blocks the airway in obstructive sleep apnea. Just before bedtime, patients place a plate with a second magnet above the throat area, which opens up the airway. The Magnap group is currently recruiting patients for a clinical trial.
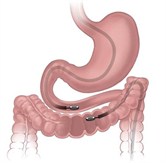 Magnamosis: Harrison has performed thousands of anastomoses, stitching together two previously unconnected tubes – for example, sewing together two pieces of the large intestine to create a detour around a bowel obstruction. Harrison and his team have designed a pair of coin-sized magnets which can create an anastomosis less invasively. Using a minimally invasive procedure, the surgeon places the magnets on the inside of each of the “loose ends” the surgeon wants to connect. The magnets snap together and cut off blood supply to the compressed tissue, which dies.
Magnamosis: Harrison has performed thousands of anastomoses, stitching together two previously unconnected tubes – for example, sewing together two pieces of the large intestine to create a detour around a bowel obstruction. Harrison and his team have designed a pair of coin-sized magnets which can create an anastomosis less invasively. Using a minimally invasive procedure, the surgeon places the magnets on the inside of each of the “loose ends” the surgeon wants to connect. The magnets snap together and cut off blood supply to the compressed tissue, which dies.
“The necrosed tissue and magnets fall inside the bowel about five days later, get pooped out, and you’ve got a new connection,” said Harrison. “The magnets have a very cool, patented secret: the shape of the magnets’ mating surfaces is not a flat-to-flat connection, but instead has a very subtle curve. The compression rots out the tissue in the middle of the magnets, but not the outside of the [circle], which heals and forms a healthy connection.” The team recently performed first-in-human studies.
In addition to developing new devices and founding the field of fetal surgery, Harrison finds great satisfaction in watching young surgeons and researchers enter the field. “They come through, fall in love with it, and do great things,” he said.
On his cubicle wall, there is a framed quote by nuclear physicist Isidor I. Rabi, PhD, which sums up Harrison’s philosophy:
“You do an experiment because your own philosophy makes you want to know the result. It’s too hard, and life is too short, to spend your time doing something because someone else has said it’s important. You must feel the thing yourself.”
“It’s personal – it’s got to be important to you,” said Harrison. “That’s not hard to do when you are a pediatric surgeon. If the patient is going to die, or they’re going to have a terrible condition, that’s enough motivation.”
Harrison and his wife, Gretchen, have four grown children – including a daughter, Brittany, who is a fourth-year medical student at UCSF – and six grandchildren.
by Elizabeth Chur